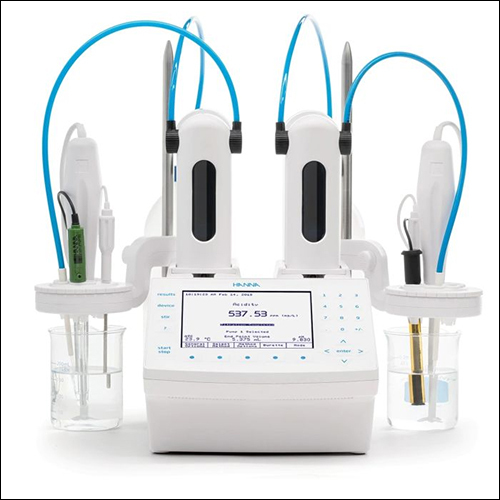
HI931 Automatic Potentiometric Titrator
Price 293550 INR/ Piece
HI931 Automatic Potentiometric Titrator Specification
- Temperature
- 0.1C; 0.1F; 0.1K Kelvin (K)
- Dimension (L*W*H)
- 315 x 205 x 375 Millimeter (mm)
- Color
- White
- Power
- 100-240 Volt (v)
- Application
- INDUSTRIAL
- Material
- Plastic
- Product Type
- HI931 Automatic Potentiometric Titrator
HI931 Automatic Potentiometric Titrator Trade Information
- Minimum Order Quantity
- 1 Piece
- Supply Ability
- 1 Piece Per Day
- Delivery Time
- 3-4 Week
About HI931 Automatic Potentiometric Titrator
SPECIFICATIONS
- Microprocessor Based
- Two Methods : FED/SVD
- FED : Fixed End Point Dosing
- SVD : Step Volume Dosing
- Three modes of Titrations :
Blank / Normality / Sample
- Automatic filling and one key stroke Rinse operation.
- PTFE Rod Stirrer with speed adjusted by knob.
- Eject facility with simultaneous display of mv or pH.
- Formulae - for Normality, %, ppm concentration, AN / TBN.
- Result Unit - %, ppm, (mg KOH/g)
- Calibration 3 pt. calibration for pH
- Tme, Date, Titration No. facility
- Reports - Result Report, Document Report, ml/mv
- Ep - 2 Eps
- Facility to enter Burette Factor for Calibration purpose
- Measuring Range - 0-14 pH, -1999 to+ 1999 mv
- Burette Volume - 20 ml.
- Burette Resolution - 0.001 ml./step
Printer interface - parallel port for Printers
Frequently Asked Questions:
1. What is the principle of potentiometric titration?
Ans. Potentiometric titration precept of potentiometry principle. Whilst the pair of electrodes are located within the pattern solution or analyte, it indicates the ability distinction between two electrodes by the addition of the titrant or by the alternate within the attention of ions.
2. How does an automatic titrator work?
Ans. These electrodes work by means of detecting the voltage trade on account of the chemical reaction on the point wherein all the analyte is taken via the titrant. The automatic titrator then analyzes the facts and determines the exact quantity at the endpoint.
3. What is potentiometry used for?
Ans. Potentiometry is a way this is used in analytical chemistry, commonly to discover the concentration of a solute in a solution. In this method, the potential among electrodes has measured with the use of a high-impedance voltmeter (wang, 2000). The use of a high-impedance voltmeter guarantees that the present flow is negligible.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Titrator Category
HI932 Automatic Potentiometric Titrator
Minimum Order Quantity : 1 Piece
Power : 100240 Watt (w)
Application : INDUSTRIAL
Color : White
Product Type : HI932 Automatic Potentiometric Titrator
HI933 Karl Fischer Volumetric Titrator for Moisture Determination
Minimum Order Quantity : 1 Piece
Power : 100240 Volt (v)
Application : INDUSTRIAL
Color : White
Product Type : HI933 Karl Fischer Volumetric Titrator for Moisture Determination
Material : Plastic
Hanna Karl Fischer Titration
Minimum Order Quantity : 1 Piece
Power : 100240 Volt (v)
Application : INDUSTRIAL
Color : White
Product Type : Hanna Karl Fischer Titration
Material : Plastic
HI934 Karl Fischer Coulometric Titrator
Minimum Order Quantity : 1 Piece
Power : 100240 Volt (v)
Application : INDUSTRIAL
Color : White
Product Type : HI934 Karl Fischer Coulometric Titrator
Material : Plastic
GST : 27AACCH2670Q1Z4
 |
HANNA EQUIPMENTS INDIA PVT. LTD.
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |


 Send Inquiry
Send Inquiry